Please site this table as follows:
Laurien De Roeck, Hiska L van der Weide, Daniëlle BP Eekers, Erik Roelofs, Esther GC Troost, Maarten Lambrecht EPTN consensus-based toxicity scoring standard for the follow-up of adult brain and base of skull tumours after radiotherapy CancerData, 2021; doi:10.17195/candat.2021.09.1
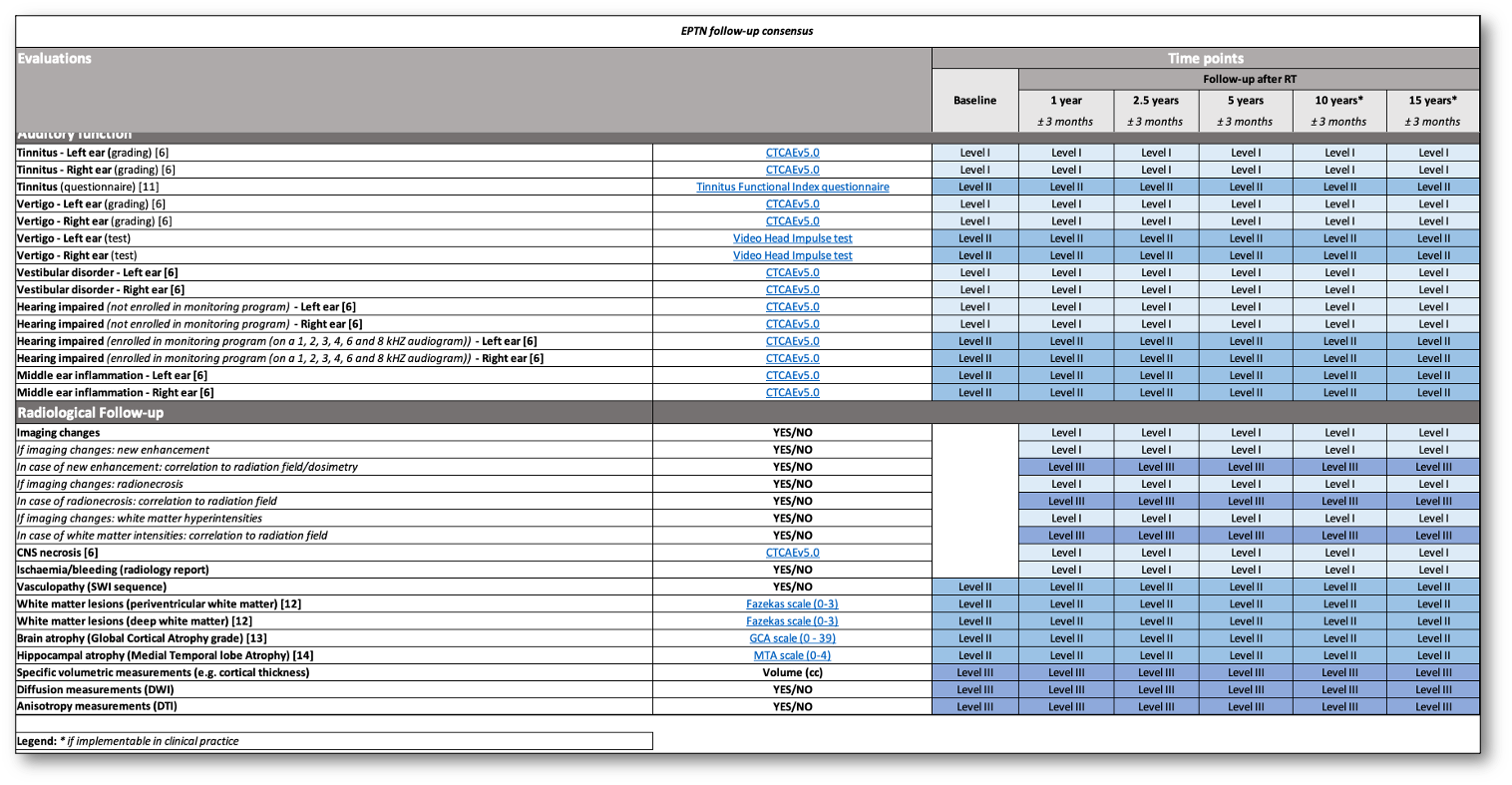
De Roeck et al. have published a guideline for consistent toxicity scoring and follow-up in adult brain tumour patients in consensus with the European Particle Therapy Network (EPTN). The purpose of this framework is to enable the collection of uniform toxicity data of brain tumour patients treated with photon and particle therapy. This will facilitate collaboration and further propel the research field of radiation-induced toxicities relevant for these patients.
In this consensus, multiple levels of recommendation are used. Level I includes all recommendations that are considered minimum of care, whereas level II and III are optional evaluations in the advanced clinical or research setting, respectively. All clinical endpoints are divided into nine outcome domains (general, hair, neurological function, neurocognitive function, endocrine function, visual pathway, ocular function, auditory function and radiological outcome), listing the proposed evaluation methods per level.
To aid in the implementation of this guideline, we hereby provide an online and interactive spreadsheet for toxicity scoring and follow-up, which can be adapted according to the feasibility and needs of each center. When required this instrument will be updated according to new insights.
| Attachment | Size |
|---|---|
| 351.96 KB | |
| 16.83 KB |