Please site this table as follows:
Eekers D, Lambrecht M, Nyström PDW, Swinnen A, Wesseling FWR, Roelofs E, Troost EG EPTN consensus-based guideline for the tolerance dose per fraction of organs at risk in the brain CancerData, 2018; doi:10.17195/candat.2018.01.1
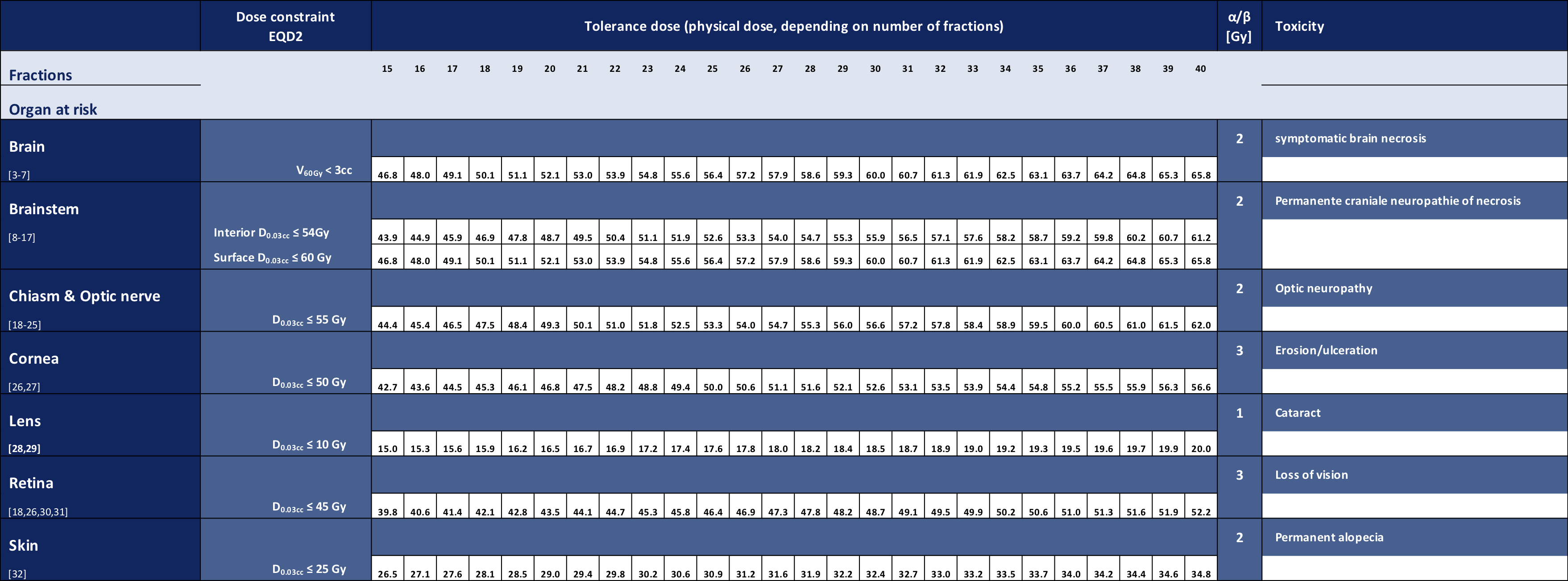
EPTN consensus-based guideline for the tolerance dose per fraction of organs at risk in the brain
In collaboration with the European Particle Therapy Network, a consensus table of tolerance doses for Organs at Risk in the brain was created. These values were all defined as a near maximum EQD2 values. Using the linear quadratic formula the doses are recalculated depending on the number of fractions.
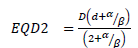
EQD2 = equivalent dose in 2Gy-fractions
D = the total dose
d = the dose per fraction
α/β = were derived from literature
Please note that this table does not include Dmean dose constraints, since these are comprised of the treatment plan's mean dose and standard deviation, which are patient- and technique-dependent (for further reading and conversion [1,2]).
(NB Downloads are below)
References:
- Hoffmann AL, Nahum AE. Fractionation in normal tissues: the (α/β)eff concept can account for dose heterogeneity and volume effects. Phys Med Biol 2013;58:6897–914. doi:10.1088/0031-9155/58/19/6897.
- Perkó Z, Bortfeld TR, Hong TS, Wolfgang J, Unkelbach J. Derivation of mean dose tolerances for new fractionation schemes and treatment modalities. Phys Med Biol 2017. doi:10.1088/1361-6560/aa9836.
- Mayo C, Yorke E, Merchant TE. Radiation associated brainstem injury. Int J Radiat Oncol Biol Phys 2010;76:S36-41. doi:10.1016/j.ijrobp.2009.08.078.
- Lawrence YR, Li XA, el Naqa I, Hahn CA, Marks LB, Merchant TE, et al. Radiation Dose–Volume Effects in the Brain. Int J Radiat Oncol 2010;76:S20–7. doi:10.1016/j.ijrobp.2009.02.091.
- Su S-F, Huang Y, Xiao W, Huang S-M, Han F, Xie C, et al. Clinical and dosimetric characteristics of temporal lobe injury following intensity modulated radiotherapy of 885 nasopharyngeal carcinoma. Radiother Oncol 2012;104:312–6. doi:10.1016/j.radonc.2012.06.012.
- Zhou X, Ou X, Xu T, Wang X, Shen C, Ding J, et al. Effect of Dosimetric Factors on Occurrence and Volume of Temporal Lobe Necrosis Following Intensity Modulated Radiation Therapy for Nasopharyngeal Carcinoma: A Case-Control Study. Int J Radiat Oncol 2014;90:261–9. doi:10.1016/j.ijrobp.2014.05.036.
- McDonald MW, Linton OR, Calley CSJ. Dose–Volume Relationships Associated With Temporal Lobe Radiation Necrosis After Skull Base Proton Beam Therapy. Int J Radiat Oncol 2015;91:261–7. doi:10.1016/j.ijrobp.2014.10.011
- Flickinger JC, Lunsford LD, Singer J, Cano ER, Deutsch M. Megavoltage external beam irradiation of craniopharyngiomas: analysis of tumor control and morbidity. Int J Radiat Oncol Biol Phys 1990;19:117–22.
- Guimas V, Thariat J, Graff-Cailleau P, Boisselier P, Pointreau Y, Pommier P, et al. [Intensity modulated radiotherapy for head and neck cancer, dose constraint for normal tissue: Cochlea vestibular apparatus and brainstem]. Cancer Radiother 2016;20:475–83. doi:10.1016/j.canrad.2016.07.077.
- Jian JJ-M, Cheng SH, Tsai SY-C, Yen K-CL, Chu N-M, Chan K-Y, et al. Improvement f local control of T3 and T4 nasopharyngeal carcinoma by hyperfractionated radiotherapy and concomitant chemotherapy. Int J Radiat Oncol Biol Phys 2002;53:344–52.
- Uy NW, Woo SY, Teh BS, Mai WY, Carpenter LS, Chiu JK, et al. Intensity-modulated radiation therapy (IMRT) 910 for meningioma. Int J Radiat Oncol Biol Phys 2002;53:1265–70.
- Schoenfeld GO, Amdur RJ, Morris CG, Li JG, Hinerman RW, Mendenhall WM. Patterns of Failure and Toxicity after Intensity-Modulated Radiotherapy for Head and Neck Cancer. Int J Radiat Oncol 2008;71:377–85. doi:10.1016/j.ijrobp.2007.10.010.
- Zheng Y, Han F, Xiao W, Xiang Y, Lu L, Deng X, et al. Analysis of late toxicity in nasopharyngeal carcinoma patients treated with intensity modulated radiation therapy. Radiat Oncol 2015;10:17. doi:10.1186/s13014-014-0326-z.
- Weber DC, Malyapa R, Albertini F, Bolsi A, Kliebsch U, Walser M, et al. Long term outcomes of patients with skull-base low-grade chondrosarcoma and chordoma patients treated with pencil beam scanning proton therapy. Radiother Oncol 2016;120:169–74. doi:10.1016/j.radonc.2016.05.011.
- Nishimura H, Ogino T, Kawashima M, Nihei K, Arahira S, Onozawa M, et al. Proton-Beam Therapy for Olfactory Neuroblastoma. Int J Radiat Oncol 2007;68:758–62. doi:10.1016/j.ijrobp.2006.12.071.
- Noël G, Feuvret L, Calugaru V, Dhermain F, Mammar H, Haie-Méder C, et al. Chordomas of the base of the skull and upper cervical spine. One hundred patients irradiated by a 3D conformal technique combining photon and proton beams. Acta Oncol (Madr) 2005;44:700–8. doi:10.1080/02841860500326257.
- Debus J, Hug EB, Liebsch NJ, O’Farrel D, Finkelstein D, Efird J, et al. Brainstem tolerance to conformal radiotherapy of skull base tumors. Int J Radiat Oncol Biol Phys 1997;39:967–75.
- Archer DB, Amoaku WMK, Gardiner TA. Radiation retinopathy—Clinical, histopathological, ultrastructural and experimental correlations. Eye 1991;5:239–51. doi:10.1038/eye.1991.39.
- McClellan RL, el Gammal T, Kline LB. Early bilateral radiation-induced optic neuropathy with follow-up MRI. Neuroradiology 1995;37:131–3.
- Jiang GL, Tucker SL, Guttenberger R, Peters LJ, Morrison WH, Garden AS, et al. Radiation-induced injury to the visual pathway. Radiother Oncol 1994;30:17–25.
- Goldsmith BJ, Rosenthal SA, Wara WM, Larson DA. Optic neuropathy after irradiation of meningioma. Radiology 1992;185:71–6. doi:10.1148/radiology.185.1.1523337.
- Martel MK, Sandler HM, Cornblath WT, Marsh LH, Hazuka MB, Roa WH, et al. Dose-volume complication analysis for visual pathway structures of patients with advanced paranasal sinus tumors. Int J Radiat Oncol Biol Phys 1997;38:273–84.
- Flickinger JC, Lunsford LD, Singer J, Cano ER, Deutsch M. Megavoltage external beam irradiation of craniopharyngiomas: analysis of tumor control and morbidity. Int J Radiat Oncol Biol Phys 1990;19:117–22.
- Hoppe BS, Stegman LD, Zelefsky MJ, Rosenzweig KE, Wolden SL, Patel SG, et al. Treatment of nasal cavity and paranasal sinus cancer with modern radiotherapy techniques in the postoperative setting—the MSKCC experience. Int J Radiat Oncol 2007;67:691–702. doi:10.1016/j.ijrobp.2006.09.023.
- Mackley HB, Reddy CA, Lee S-Y, Harnisch GA, Mayberg MR, Hamrahian AH, et al. Intensity-modulated radiotherapy for pituitary adenomas: the preliminary report of the Cleveland Clinic experience. Int J Radiat Oncol Biol Phys 2007;67:232–9. doi:10.1016/j.ijrobp.2006.08.039.
- Cancer Institute N. Common Terminology Criteria for Adverse Events (CTCAE) Common Terminology Criteria for Adverse Events v4.0 (CTCAE) 2009.
- Barabino S, Raghavan A, Loeffler J, Dana R. Radiotherapy-induced ocular surface disease. Cornea 2005;24:909–14.
- Merriam GR, Worgul B V, Worgul B V. Experimental radiation cataract--its clinical relevance. Bull N Y Acad Med 1983;59:372–92.
- Henk JM, Whitelocke RA, Warrington AP, Bessell EM. Radiation dose to the lens and cataract formation. Int J Radiat Oncol Biol Phys 1993;25:815–20.
- Brown GC, Shields JA, Sanborn 705 G, Augsburger JJ, Savino PJ, Schatz NJ. Radiation retinopathy. Ophthalmology 1982;89:1494–501.
- Parsons JT, Bova FJ, Fitzgerald CR, Mendenhall WM, Million RR. Radiation retinopathy after external-beam irradiation: analysis of time-dose factors. Int J Radiat 720 Oncol Biol Phys 1994;30:765–73.
- Stram DO, Mizuno S. Analysis of the DS86 atomic bomb radiation dosimetry methods using data on severe epilation. Radiat Res 1989;117:93–113.
| Attachment | Size |
|---|---|
| 88.83 KB | |
| 20.36 KB | |
| 34.34 KB |