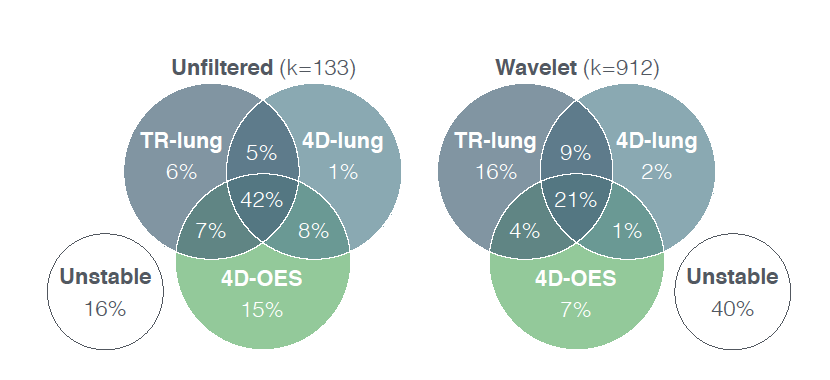
Venn chart visualizing the overlap of stable features with CCC\>0.85 in the test-retest lung (TR-lung), 4D-lung and 4D-oesophagus (4D-OES) dataset respectively.
Background and Purpose
Patients with low-grade glioma (LGG) have a prolonged survival expectancy due to better discriminative tumor classification and multimodal treatment. Consequently, long-term treatment toxicity gains importance. Contemporary radiotherapy techniques such as intensity-modulated radiotherapy (IMRT), volumetric modulated arc therapy (VMAT), Tomotherapy (TOMO) and intensity-modulated proton therapy (IMPT) enable high-dose irradiation of the target but they differ regarding delivered dose to organs at risk (OARs). The aim of this comparative in silico study was to determine these dosimetric differences in delivered doses.
Material and Methods
Imaging datasets of twenty-five LGG patients having undergone postoperative radiotherapy were included. For each of these patients, in silico treatment plans to a total dose of 50.4Gy to the target volume were generated for the four treatment modalities investigated (i.e., IMRT, VMAT, TOMO, IMPT). Resulting treatment plans were analyzed regarding dose to target and surrounding OARs comparing IMRT, TOMO and IMPT to VMAT.
Results
In total 100 treatment plans (4 per patient) were analyzed. Compared to VMAT the IMPT mean dose (Dmean) for 9 out of 10 (90\%) OARs was statistically significantly (p\<0.02) reduced, for TOMO this was true in 3/10 (30\%) patients and for 1/10 (10\%) patients for IMRT. IMPT was the prime modality reducing dose to the OARs followed by TOMO.
Discussion
The low dose volume to the majority of OARs was significantly reduced when using IMPT compared to VMAT. Whether this will lead to a significant reduction in neurocognitive decline and improved quality of life is to be determined in carefully designed future clinical trials.
}, doi = {10.17195/candat.2018.05.1}, author = {Danielle Eekers and Erik Roelofs and Macarena Cubillos-Mesias and Charles Ni{\"e}l and Robert Jan Smeenk and Ann Hoeben and Andre W.H. Minken and Johannes H.A.M. Kaanders and Philippe Lambin and Esther G.C. Troost} } @mastersthesis {98, title = {Optimization of Brain and Head \& Neck Radiotherapy}, volume = {Ph.D.}, year = {2018}, month = {10/2018}, pages = {201}, school = {Maastricht University}, address = {Maastricht}, abstract = {The aim of this thesis is to further optimise radiation therapy of Brain and Head \& Neck by reducing the dose to the healthy surrounding tissue, so called organs at risk (OARs), leading to a reduction in side effects.
The first objective of this doctoral thesis was to assess the value of proton therapy in reducing the dose to the OARs, in particular for re-irradiation in head and neck squamous cell carcinoma and primary irradiation of low-grade glioma. Chapters 2 and 3 report on two in silico trials conducted within the international Radiation Oncology Collaborative Comparison (ROCOCO) consortium, comparing different radiotherapy modalities including proton therapy, to assess the potential gains for individual patients due to the dosimetric characteristics of particle therapy. The first trial compared intensity-modulated proton therapy (IMPT) and carbon-ion therapy (IMIT) with the golden standard volumetric modulated arc therapy (VMAT) when re-irradiating patients with head and neck squamous cell carcinoma. The second trial compared intensity- modulated radiation therapy (IMRT), IMPT and helical tomotherapy (TOMO) with the golden standard VMAT in patients with a low-grade glioma. Both trials demonstrated that particle therapy can significantly reduce the dose to OARs whilst maintaining the prescription dose. Whether this translates into a clinically relevant benefit, is the subject of future research. In order to predict such a benefit, normal tissue complication probability (NTCP) models are needed. Validated NTCP models are currently lacking for head and neck re-irradiation as well as for the primary treatment of the central nervous system (CNS).
In Chapter 4, the posterior cerebellum is introduced as a new, potentially relevant OAR for the future development of an NTCP model that is focused on cognition, based on the growing evidence from structural and functional imaging studies that the cerebellum plays a role in neurocognition.
Delineation of the relevant OARs on computed tomography (CT) and magnetic resonance imaging (MRI) is needed to optimise the treatment plan before administering the corresponding dose to the patient. This manual delineation is a time- consuming process and a well-known source of error within the planning process, due to inter- and intra-observer variability1.
Reducing this CNS OAR delineation variability is the second objective of this thesis, as described in Chapter 5. An international group of expert radiation oncologists in the field of neuro-oncology reached agreement on the European Particle Therapy Network\ (EPTN) consensus-based CNS delineation atlas, in order to decrease the variation in CNS delineation. This CNS atlas is presented online (www.cancerdata.org) and encompasses delineation instructions for 15 CNS OARs, including the posterior cerebellum. It includes one CT-scan at two different brightness and contrast settings and two MR scans (3 and 7 Tesla) showing the OARs in three directions (axial, coronal and sagittal view). In Chapter 6, an EPTN consensus-based normal tissue tolerance table, including all currently known and deemed relevant CNS OARs, reports the tolerance dose in equivalent dose (EQD2), which enables a uniform comparison of different treatment modalities in the future (www.cancerdata.org). The use of the consensus-based EPTN CNS atlas and tolerance table is recommended for the Dutch model-based approach comparing photon and proton beam irradiation as well as for future prospective clinical trials including novel radiation techniques and/or modalities2.
The third objective of this thesis was determining the role of radiotherapy in the treatment of epilepsy. Chapter 7 contains a systematic review about the evidence on the efficacy of primary radiosurgery or stereotactic radiotherapy for drug-resistant non- neoplastic focal epilepsy in adults. After treatment, an average of 58\% of the patients reported no or rare seizures (defined as radiotherapy-adapted Engel class [RAEC] I and II). A dose-effect model was fitted to the available response data to derive a relationship between prescribed dose and RAEC frequency of this, in the Netherlands, new indication for radiotherapy. In Chapter 8, the results of the previous mentioned chapters of this thesis are being discussed and future perspectives are presented. Additional research needs to be conducted to gain further knowledge to fully understand the potential of particle therapy. Thanks to solid collaborations throughout the radiotherapy community and beyond, with all our colleagues in the medical field, there is a unique possibility to further optimise the treatment of Brain and Head \& Neck together.
References
Vinod SK, Jameson MG, Min M, Holloway LC. Uncertainties in volume delineation in radiation oncology: A systematic review and recommendations for future studies. Radiother. Oncol 2016;121:169{\textendash}79.
Widder J, Schaaf van der A, Lambin P, et al. The quest for evidence for proton therapy: the model-based\ approach and precision medicine. Int J Radiat Oncol Biol Phys 2015. pii: S0360-3016:26569-8.\
Eekers et al. have recently proposed a neuro-oncology atlas [doi:10.17195/candat.2017.08.1, Ed.], which was co-authored by most centers associated in the European Proton Therapy Network (EPTN). With the introduction of new treatment techniques, such as integrated magnetic resonance imaging and linear accelerators (MR-linac) or particle therapy, the prediction of clinical efficacy of these more costly treatment modalities becomes more relevant. One of the side-effects of brain irradiation, being cognitive decline, is one of the toxicities most difficult to measure and predict. In order to validly compare different treatment modalities, 1) a uniform nomenclature of the organs at risk (OARs), 2) uniform atlas-based delineation [e.g., Eekers et al.], 3) long-term follow-up data with standardized cognitive tests, 4) a large patient population, and 5) (thus derived) validated normal tissue complication probability (NTCP) models are mandatory.
Apart from the Gondi model, in which the role of the dose to 40\% of both hippocampi (HC) proves to be significantly related to cognition in 18 patients, no similar models are available. So there is a strong need for more NTCP models, on HC, brain tissue and possible other relevant brain structures.
In this review we summarize the available evidence on the role of the posterior cerebellum as a possible new organ at risk for cognition, which is deemed relevant for irradiation of brain and head and neck tumors.
}, keywords = {Cognition, Delineation atlas, Neuro-oncology, Organ at risk, Posterior cerebellum, Radiotherapy}, issn = {24056308}, doi = {10.1016/j.ctro.2017.11.010}, url = {http://linkinghub.elsevier.com/retrieve/pii/S2405630817300769}, author = {Eekers, Dani{\"e}lle B.P. and Lieke in {\textquoteright}t Ven and Deprez, Sabine and Jacobi, Linda and Erik Roelofs and Ann Hoeben and Lambin, Philippe and Dirk De Ruysscher and Troost, Esther G.C.} } @article {80, title = {Data from: 4DCT imaging to assess radiomics feature stability: an investigation for thoracic cancers}, year = {2017}, publisher = {CancerData}, abstract = {Background and Purpose
Quantitative tissue characteristics derived from medical images, also called radiomics, contain valuable prognostic information in several tumour-sites. The large number of features available increases the risk of overfitting. Typically test-retest CT-scans are used to reduce dimensionality and select robust features. However, these scans are not always available. We propose to use different phases of respiratory-correlated 4D CT-scans (4DCT) as alternative.\
Materials and Methods
In test-retest CT-scans of 26 non-small cell lung cancer (NSCLC) patients and 4DCT-scans (8 breathing phases) of 20 NSCLC and 20 oesophageal cancer patients, 1045 radiomics features of the primary tumours were calculated. A concordance correlation coefficient (CCC) \>0.85 was used to identify robust features. Correlation with prognostic value was tested using univariate cox regression in 120 oesophageal cancer patients.
Results
Features based on unfiltered images demonstrated greater robustness than wavelet-filtered features. In total 63/74 (85\%) unfiltered features and 268/299 (90\%) wavelet features stable in the 4D-lung dataset were also stable in the test-retest dataset. In oesophageal cancer 397/1045 (38\%) features were robust, of which 108 features were significantly associated with overall-survival.
Conclusion
4DCT-scans can be used as alternative to eliminate unstable radiomics features as first step in a feature selection procedure. Feature robustness is tumour-site specific and independent of prognostic value.
\

Venn chart visualizing the overlap of stable features with CCC\>0.85 in the test-retest lung (TR-lung), 4D-lung and 4D-oesophagus (4D-OES) dataset respectively.
Purpose
Tools for survival prediction for non-small cell lung cancer (NSCLC) patients treated with (chemo)radiotherapy are of limited quality. In this work, we develop a predictive model of survival at two years based on a large volume of historical patient data, as a proof of concept, using a distributed learning approach.
Patients and methods
Clinical data from 698 lung cancer patients, treated with curative intent with chemoradiation (CRT) or radiotherapy (RT) alone were collected and stored in 2 different cancer institutes (559 patients at Maastro clinic (Netherlands), 139 at University of Manchester (UK). The model was further validated on 196 patients originating from the University of Michigan (USA).
A Bayesian network model is adapted for distributed learning (watch the animation). Two-year post-treatment survival was chosen as endpoint.\ The Institute 1 cohort data is publicly available\ and the developed models can be found at\ PredictCancer.org)
Results
Variables included in the final model were T and N stage, age, performance status, and total tumor dose. The model has an AUC of 0.66 on the external validation set and an AUC of 0.62 on a 5-fold cross-validation. A model based on T and N stage performed with an AUC of 0.47 on the validation set, significantly worse than our model (P\<0.001). A high- and low-risk chance of survival group can be identified using the model presented in this study, these groups have significantly different overall survival (P\<0.01).
Conclusion
Distributed learning from federated databases allows learning of predictive models on data originating from multiple institutions while avoiding many of the data sharing barriers.\ We believe that Distributed learning is the future of sharing data in health care.
}, keywords = {Bayesian network, lung cancer, NSCLC, prediction model}, doi = {10.17195/candat.2017.02.2}, author = {Arthur Jochems and Timo M. Deist and Issam El Naqa and Marc Kessler and Chuck Mayo and Jackson Reeves and Shruti Jolly and Martha Matuszak and Randall Ten Haken and Johan van Soest and Cary Oberije and Corinne Faivre-Finn and Gareth Price and Dirk De Ruysscher and Philippe Lambin and Andr{\'e} Dekker} } @article {77, title = {Data from: Survival prediction of non-small cell lung cancer patients using radiomics analyses of cone-beam CT images}, year = {2017}, abstract = {Background and Purpose: In this study we investigated the interchangeability of planning CT and cone-beam CT (CBCT) extracted radiomic features. Furthermore, this study validates a previously\ described CT based prognostic radiomic signature for non-small cell lung cancer (NSCLC) patients using CBCT based features.
Material and Methods: One internal dataset of 132 and two external datasets of 62 and 94 stage I-IV NSCLC patients were included in this study. Interchangeability was assessed by performing a linear regression on CT and CBCT extracted features. A two-step correction was applied prior to model validation of a previously published radiomic signature.
Results: 13.3\% (149 out of 1119) of the radiomic features, including all features of the previously published radiomic signature, showed an R2 above 0.85 between intermodal imaging techniques. For the radiomic signature, Kaplan-Meier curves were significantly different between groups with high and low prognostic value for both modalities. Harrell{\textquoteright}s concordance index was 0.69 for CT and 0.66 for CBCT models for dataset 1.
Conclusions: The results show that a subset of radiomic features extracted from CT and CBCT images are interchangeable using simple linear regression. Moreover, a previously developed radiomics signature has prognostic value for overall survival in three CBCT cohorts, showing the potential of CBCT radiomics to be used as prognostic imaging biomarker.
\
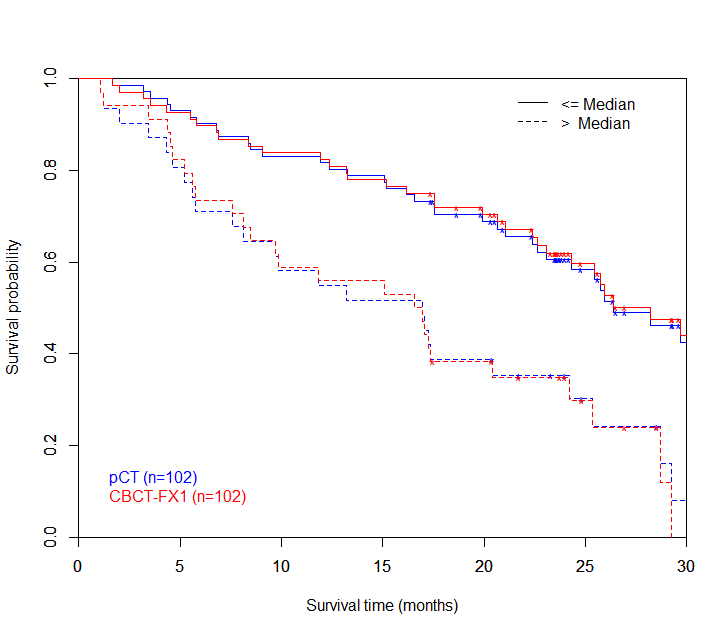
Kaplan-Meier curves for pCT and CBCT. Kaplan-Meier curves are based on model predictions of the radiomic signature.
}, keywords = {computed tomography, cone-beam CT, non-small cell lung cancer, Radiomics, survival prediction}, doi = {10.17195/candat.2017.02.1}, author = {Janita van Timmeren and Ralph T.H. Leijenaar and Wouter van Elmpt and Bart Reymen and Cary Oberije and Ren{\'e} Monshouwer and Johan Bussink and Carsten Brink and Olfred Hansen and Philippe Lambin} } @article {111, title = {Developing and Validating a Survival Prediction Model for NSCLC Patients Through Distributed Learning Across 3 Countries}, journal = {International Journal of Radiation Oncology*Biology*Physics}, volume = {99}, year = {2017}, month = {Jan-10-2017}, pages = {344 - 352}, issn = {03603016}, doi = {10.1016/j.ijrobp.2017.04.021}, url = {https://linkinghub.elsevier.com/retrieve/pii/S0360301617308258https://api.elsevier.com/content/article/PII:S0360301617308258?httpAccept=text/xmlhttps://api.elsevier.com/content/article/PII:S0360301617308258?httpAccept=text/plain}, author = {Jochems, Arthur and Deist, Timo M. and El Naqa, Issam and Kessler, Marc and Mayo, Chuck and Reeves, Jackson and Jolly, Shruti and Matuszak, Martha and Ten Haken, Randall and van Soest, Johan and Oberije, Cary and Faivre-Finn, Corinne and Price, Gareth and De Ruysscher, Dirk and Lambin, Philippe and Dekker, Andre} } @article {65, title = {Data from: Benefit of particle therapy in re-irradiation of head and neck patients. Results of a multicenter in silico ROCOCO trial}, year = {2016}, abstract = {Background and Purpose
In this multicentric in silico trial we compared photon, proton, and carbon ion radiotherapy plans for re-irradiation of patients with squamous cell carcinoma of the head and neck (HNSCC) regarding dose to tumour and doses to surrounding organs at risk (OARs).
Material and Methods
Twenty-five HNSCC patients with a second new or recurrent cancer after previous irradiation (70 Gy) were included. Intensity-modulated proton therapy (IMPT) and ion therapy (IMIT) re-irradiation plans to, again, a total dose of 70 Gy were compared to photon therapy delivered with volumetric modulated arc therapy (VMAT).
Results
When comparing IMIT and IMPT to VMAT the mean dose to all 22 OARs was statistically significantly reduced for IMIT and to 15 out of 22 OARs (68\%) using IMPT. The maximum dose to 2\% volume (D2) of the brainstem and spinal cord were statistically significantly reduced using IMPT and IMIT compared to VMAT. The data are available on www.cancerdata.org.
Conclusions
In this ROCOCO in silico trial, a reduction in mean dose to OARs was achieved using particle therapy compared to photons in the re-irradiation of HNSCC. There was a dosimetric benefit favouring carbon-ions above proton therapy. These dose reductions may potentially translate into lower severe complication rates related to the re-irradiation.
}, doi = {10.17195/candat.2016.04.2}, author = {Danielle Eekers and Erik Roelofs and Urszula Jelen and Maura Kirk and Marlies Granzier and Filippo Ammazzalorso and Peter H. Ahn and Geert O.R.J Janssens and Frank Hoebers and Tobias Friedmann and Timothy Solberg and Sean Walsh and Esther G C Troost and Johannes H.A.M. Kaanders and Philippe Lambin} } @article {72, title = {Data from: Nitroglycerin in non-small cell lung cancer: does it impact tumor hypoxia and tumor perfusion? A window-of-opportunity clinical trial.}, year = {2016}, abstract = {Despite preclinical evidence that nitric oxide (NO) donors influence both tumor perfusion and hypoxia, in clinical trials nitroglycerin has not been shown to improve the treatment results of all patients with non-small cell lung cancer (NSCLC). Biomarkers are therefore needed to select patients for treatment with NO donors. In this window-of-opportunity study we demonstrate the effect of nitroglycerin on hypoxia in patients using repeated hypoxia PET-imaging: we observed a reduction of hypoxia - quantified by uptake of PET tracer HX4- of varying magnitude upon application of a nitroglycerin patch in approximately two thirds of patients with hypoxic loco-regional disease.
In a quarter of patients with baseline hypoxia, the tumor and nodes become normoxic after treatment with nitroglycerin. Furthermore, through dynamic contrast enhanced CT-scans (DCE-CT) we demonstrated that this effect on hypoxic tracer uptake is negatively correlated with tumor perfusion in hypoxic tumors.
Using in vitro experiments we show that the metabolism of 2-nitroimidazoles is not influenced by nitroglycerin itself and furthermore that nitroglycerin does not inhibit mitochondrial respiration at human achievable concentrations. Therefore, 2-nitroimidazole based imaging biomarkers can be used to evaluate the nitroglycerin treatment efficacy in an individual patient.\
The current results do support the hypothesis that hypoxia scans and/or DCE-CT scans could form a tool to select patients for a nitroglycerin patch adjuvant to anti-cancer treatment (radiotherapy, chemotherapy, targeted agents or immunotherapy) to increase drug uptake and/or to decrease hypoxia.
An animation summarizing our results is available at https://youtu.be/udJSBYaRv9w.
\
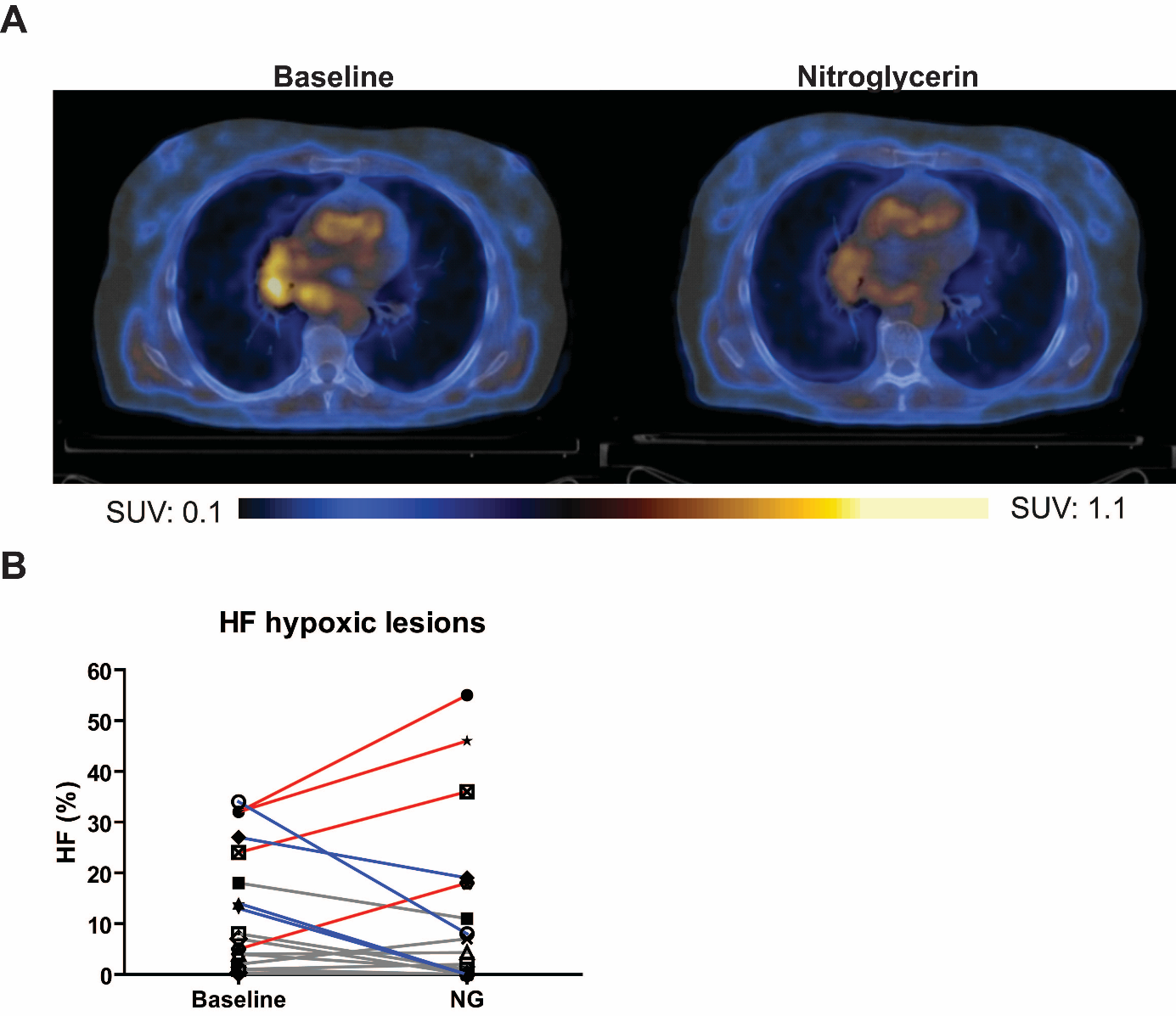
Figure 1. Uptake of HX4 before and after nitoglycerin administration
A. Images of a HX4 PET/CT scan before and after nitroglycerin administration of the same patient.
B. Evolution of high-uptake fraction (HF) of HX4 after application of nitroglycerin patch in baseline hypoxic lesions. Blue = decline \> CR of HX4 (17\%), grey = stable: change \< CR.
\
Note: dataset was updated 2017-11-24}, doi = {10.17195/candat.2016.07.2}, author = {Bart Reymen and Marike W. van Gisbergen and Aniek J.G. Even and Catharina M.L. Zegers and Cary Oberije and Felix Mottaghy and Marco Das and Ala Yaromina and Wouter van Elmpt and Dirk De Ruysscher and Ludwig Dubois and Philippe Lambin} } @article {64, title = {Data from: Prognostic value of blood-biomarkers related to hypoxia, inflammation, immune response and tumour load in non-small cell lung cancer {\textendash} a survival model with external validation}, year = {2016}, abstract = {
Aim: Improve the prognostic prediction of clinical variables for non-small cell lung cancer (NSCLC), by selecting from blood-biomarkers, non-invasively describing hypoxia, inflammation and tumour load.
Methods: \ Model development and validation included 182 and 181 inoperable stage I-IIIB NSCLC patients treated radically with radiotherapy (55.2\%) or chemo-radiotherapy (44.8\%). Least absolute shrinkage and selection operator (LASSO), selected from blood-biomarkers related to hypoxia [osteopontin (OPN) and carbonic anhydrase IX (CA-IX)], inflammation [interleukin-6 (IL-6), IL-8, and C-reactive protein (CRP)], and tumour load [carcinoembryonic antigen (CEA), and cytokeratin fragment 21-1 (Cyfra 21-1)]. Sequent model extension selected from alpha-2-macroglobulin (α2M), serum interleukin-2 receptor (sIL2r), toll-like receptor 4 (TLR4), and vascular endothelial growth factor (VEGF). Discrimination was reported by concordance-index.
Results: OPN and Cyfra 21-1 (hazard ratios of 3.3 and 1.7) significantly improved a clinical model comprising gender, World Health Organization performance-status, forced expiratory volume in 1 second, number of positive lymph node stations, and gross tumour volume, from a concordance-index of 0.66 to 0.70 (validation = 0.62 and 0.66). Extension of the validated model yielded a concordance-index of 0.67, including α2M, sIL2r and VEGF (hazard ratios of 4.6, 3.1, and 1.4).
Conclusion: Improvement of a clinical model including hypoxia and tumour load blood-biomarkers was validated. New immunological markers were associated with overall survival. Data and models can be found at www.cancerdata.org\ (DOI:10.17195/candat.2016.04.1) and www.predictcancer.org.
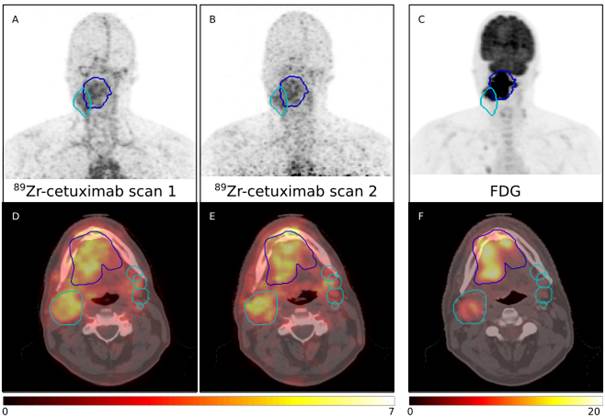
}, doi = {10.17195/candat.2016.04.1}, author = {Sara Carvalho and Esther G C Troost and Judith Bons and Paul Menheere and Philippe Lambin and Cary Oberije} } @article {75, title = {Data from: Quantitative assessment of Zirconium-89 labeled cetuximab using PETCT imaging in patients with advanced head and neck cancer - a theragnostic approach}, year = {2016}, abstract = {Biomarkers predicting treatment response to the monoclonal antibody cetuximab in locally advanced head and neck squamous cell carcinomas (LAHNSCC) are lacking. We hypothesize that tumor accessibility is an important factor in treatment success of the EGFR targeting drug. We quantified uptake of cetuximab labeled with Zirconium-89 (89Zr) using PET/CT imaging.
Seventeen patients with stage III-IV LAHNSCC received a loading dose unlabeled cetuximab, followed by 10 mg 54.5{\textpm}9.6 MBq 89Zr-cetuximab. PET/CT images were acquired either 3 and 6 or 4 and 7 days post-injection. 89Zr-cetuximab uptake was quantified using standardized uptake value (SUV) and tumor-to-background ratio (TBR), and correlated to EGFR immunohistochemistry. TBR was compared between scan days to determine optimal timing.
Uptake of 89Zr-cetuximab varied between patients (day 6-7: SUVpeak range 2.5-6.2). TBR increased significantly (49{\textpm}28\%, p\<0.01) between first (1.1{\textpm}0.3) and second scan (1.7{\textpm}0.6). Between groups with a low and high EGFR expression a significant difference in SUVmean (2.1 versus 3.0) and SUVpeak (3.2 versus 4.7) was found, however, not in TBR. Data is available at www.cancerdata.org.
In conclusion, 89Zr-cetuximab PET imaging shows large inter-patient variety in LAHNSCC and provides additional information over FDG-PET and EGFR expression. Validation of the predictive value is recommended with scans acquired 6-7 days post-injection.
Examples scans for four patients are provided. All patients received a planning PET/CT and two 89Zr-cetuximab scans. \ Patient P0037C0006I4475579 and P0037C0006I5879176 underwent 89Zr-cetuximab scans at 4 and 7 days post-injection; patient P0037C0006I6042760 and P0037C0006I8991415 at 3 and 6 days post-injection. All 89Zr-cetuximab PET scans are corrected for attenuation, scatter and 89Zr decay.
Radiation oncology is in trouble. In fact, healthcare in general is. The spending on healthcare is rapidly rising, leading to a non-sustainable situation [1]. An aging population can partly explain the increasing costs, but overtreatment plays a large role as well [2]. This particularly happens due to defensive healthcare [3]. On the other hand, overtreatment also occurs due to the longevity of clinical protocols, which can cause inefficiency, as does the technology-push from healthcare industry.
One of the options to reduce the costs is to apply cost-effectiveness analyses or Healthcare Technology Assessments to inform policy and decision makers about the rightful use of (new) technology.
To this end, the development of personalized decision support systems was investigated, including cost-effectiveness in the field of proton therapy. The following hypotheses were addressed and affirmed:
◆\ \ \ \ Rapid Learning Healthcare (RLHC) can improve clinical research and decision-making.
◆\ \ \ \ Advances in Healthcare Information Technology (HIT) enable international data sharing while preserving patient privacy.
◆\ \ \ \ Combining in silico planning comparison studies with multifactorial prediction models enables cost-effective application of limited, expensive resources, as introduced for proton therapy.
The basic concept of this thesis is the use of RLHC to complement evidence-based medicine with prediction modelling to provide clinical decision support for individualised cancer care (Chapter 2). RLHC applies advanced HIT to aggregate disparate clinical information sources into a data warehouse that presents a complete, integrated disease-oriented view of medical data for research purposes. This chapter presents a general overview of some of the techniques that were used in the following work.
Chapter 3 demonstrates how institutions can benefit from this improved presentation of patient information to improve efficiency and quality of data collection for clinical trials. Furthermore, RLHC uses machine learning principles to mine the data for clinically relevant relations to build data-driven complication prediction models.
Large amounts of data are needed for the modelling and validation of these models, which is unavailable from single institutions. Furthermore, to increase information heterogeneity, which improves the predictive value of models, and to allow for external validation it is imperative that foreign data is available.
Chapter 4 presents the necessary data-sharing techniques that unlock the medical data silos in a privacy-preserving manner by installing {\textquoteleft}connectors{\textquoteright} inside the hospital{\textquoteright}s network firewalls. Furthermore, by transforming the data into internationally standardised semantic interoperable data models, computer systems are able to translate data into knowledge. Furthermore, medical records in local languages are converted into international, unique codes. This means that the data can be reused in foreign countries with equal meaning and value, which allows for federated or distributed learning of complication prediction models [4].
In advance of a federated network, which is very advanced but not common practice yet, the framework of MISTIR (mistir.info) uses a centralised research database for international collaborative in silico clinical trials (Chapter 5). The system offers a secure data-exchange platform with high-quality datasets and trial protocols for multiple in silico planning comparisons of different treatment modalities. Quality assurance measures and automated data extraction procedures are applied for uniform analysis of the results.
Chapter 6 presents a ROCOCO (mistir.info/rococo) lung cancer trial that used the platform to investigate whether proton therapy can reduce dose to normal tissue, with equal or higher tumour dose. The trial participants downloaded de-identified clinical datasets to plan according to a strict protocol. The treatment planning results were returned to the server after which central analysis was performed. It showed that dose reduction is indeed possible, even when escalating the dose to the tumour.
Chapter 7 evaluates an online proton therapy decision support system (PRODECIS: prodecis.nl), reusing head and neck cancer data from another ROCOCO trial. The system assesses healthcare reimbursement eligibility for model-based tumour groups by comparison of photon and proton treatment plans, using publicly available toxicity and cost-prediction models from PredictCancer (predictcancer.org). The system proved successful in the assessment of 92\% of the test cases and showed that, with proton therapy, 91\% of the patients clinically benefit from reduced complications after one year, while 35\% would be considered cost-effective.
1.\ \ \ \ \ CBS. Zorguitgaven stijgen met 1,8 procent in 2014. www.cbs.nl/nl-NL/menu/themas/gezondheid-welzijn/publicaties/artikelen/archief/2015/zorguitgaven-stijgen-met-1-8-procent-in-2014.htm [accessed November 8, 2015]
2.\ \ \ \ \ Patel TY. It Is Overtreatment, Not Overdiagnosis. Acad Radiol 2015;22(8):1044{\textendash}1045. doi:10.1016/j.acra.2015.06.006
3.\ \ \ \ \ Sekhar Ms, Vyas N. Defensive medicine: A bane to healthcare. Ann Med Health Sci Res 2013;3(2):295. doi:10.4103/2141-9248.113688
4.\ \ \ \ \ Damiani A, Vallati M, Gatta R, Dinapoli N, Jochems A, Deist T, et al. Distributed Learning to Protect Privacy in Multi-centric Clinical Studies. In: Holmes JH, Bellazzi R, Sacchi L, Peek N, editors. Artificial Intelligence in Medicine, Springer International Publishing; 2015
\
\
\
}, keywords = {data, decision support, dissertation, PRODECIS, proton therapy, ROCOCO}, isbn = {978 94 6159 577 5}, doi = {10.17195/candat.2016.07.01}, author = {Erik Roelofs} } @article {73, title = {Radiomics Digital Phantom}, year = {2016}, publisher = {CancerData}, abstract = {The rise of radiomics, the high-throughput mining of quantitative image features from (standard-of-care) medical imaging for knowledge extraction and application within clinical decision support systems to improve diagnostic, prognostic, and predictive accuracy, has significant and substantial implications for the medical community. Radiomic analysis exploits sophisticated image analysis tools and the exponential growth of medical imaging data to develop and validate powerful image-based signatures/models for precision diagnosis and treatment in medicine.
This review describes the process of radiomics, its pitfalls, challenges, opportunities, and its capacity to improve clinical decision making (presently primarily in the care of patients with cancer, however, all imaged patients may benefit from quantitative radiology).
Finally, the field of radiomics is emerging rapidly; however, the field lacks standardized evaluation of both the scientific integrity and the clinical significance of the numerous published radiomics investigations resulting from this growth. There is a clear and present need for rigorous evaluation criteria and reporting guidelines in order for radiomics to mature as a discipline. We therefore provide guidance together with a novel metric, the radiomics quality score (RQS) and an online digital phantom (DOI:10.17195/candat.2016.08.1), to meet this urgent need for both past and future investigations in the field of radiomics.\ \ \
}, keywords = {CT, phantom, quantitative features, Radiomics, standardization}, doi = {10.17195/candat.2016.08.1}, author = {Philippe Lambin} } @article {61, title = {Data from: A validated Tumor Control Probability model based on a meta-analysis of low, intermediate, and high-risk prostate cancer patients treated by photon, proton, or carbon-ion radiotherapy}, year = {2015}, publisher = {CancerData}, abstract = {This dataset supports the publication by Walsh et al, entitled "A validated Tumor Control Probability model based on a meta-analysis of low, intermediate, and high-risk prostate cancer patients treated by photon, proton, or carbon-ion radiotherapy".
}, doi = {10.17195/candat.2015.10.8}, author = {Sean Walsh and Erik Roelofs and Peter Kuess and Philippe Lambin and Bleddyn Jones and Dietmar Georg and Frank Verhaegen} } @article {57, title = {Data from: Development and Evaluation of an Online Three-Level Proton vs Photon Decision Support Prototype for Head and Neck Cancer - Comparison of Dose, Toxicity and Cost-Effectiveness}, year = {2015}, month = {10/2015}, publisher = {CancerData}, abstract = {This dataset supports the publication by Cheng et al, entitled "Development and Evaluation of an Online Three-Level Proton vs Photon Decision Support Prototype for Head and Neck Cancer - Comparison of Dose, Toxicity and Cost-Effectiveness". It\ was used to evaluate the PRODECIS decision support prototype\ for online evaluation of eligiblilty for proton therapy using Dutch guidelines.\
The Head and Neck test dataset was based on the ROCOCO dataset by HP van der Laan, as described in doi:10.3109/0284186X.2012.692885.
}, keywords = {cancer, decision support, model-based, oncology, proton therapy, radiotherapy planning}, doi = {10.17195/candat.2015.10.5}, url = {https://www.cancerdata.org/resource/doi:10.17195/candat.2015.10.5}, author = {Qing Cheng and Erik Roelofs and Bram Ramaekers and Danielle Eekers and Johan van Soest and Tim Lustberg and Frank Hoebers and Hans Paul van der Laan and Erik Korevaar and Andr{\'e} Dekker and Johannes Langendijk and Philippe Lambin} } @article {60, title = {Data from: MMP2 small immuno protein antibody uptake in xenograft tumors is associated with MMP2 activity}, year = {2015}, publisher = {CancerData}, abstract = {This dataset supports the publication by\ Kranthi Panth, et al.\ In this study, it is\ hypothesized that aMMP2-SIP, a small immuno protein antibody targeting the catalytic domain of human MMP2 distinctively detects MMP2 in the tumors.\ MMP2 plays a vital role in tumorigenesis, angiogenesis and tumor invasion.
}, keywords = {integrin, matrix metalloproteinase, MMP2 activity, preclinical imaging}, doi = {10.17195/candat.2015.10.6}, url = {https://www.cancerdata.org/resource/doi:10.17195/candat.2015.10.6}, author = {Kranthi Panth and Twan van den Beucken and Rianne Biemans and Natasja Lieuwes and Marcel Weber and Mario Losen and Ala Yaromina and Ludwig Dubois and Philippe Lambin} } @article {55, title = {PET-based dose painting in non-small cell lung cancer: Comparing uniform dose escalation with boosting hypoxic and metabolically active sub-volumes.}, journal = {Radiother Oncol}, volume = {116}, year = {2015}, month = {2015 Aug}, pages = {281-6}, abstract = {BACKGROUND AND PURPOSE: We compared two imaging biomarkers for dose-escalation in patients with advanced non-small cell lung cancer (NSCLC). Treatment plans boosting metabolically active sub-volumes defined by FDG-PET or hypoxic sub-volumes defined by HX4-PET were compared with boosting the entire tumour.
MATERIALS AND METHODS: Ten NSCLC patients underwent FDG- and HX4-PET/CT scans prior to radiotherapy. Three isotoxic dose-escalation plans were compared per patient: plan A, boosting the primary tumour (PTVprim); plan B, boosting sub-volume with FDG >50\% SUVmax (PTVFDG); plan C, boosting hypoxic volume with HX4 tumour-to-background >1.4 (PTVHX4).
RESULTS: Average boost volumes were 507 {\textpm} 466 cm(3) for PTVprim, 173 {\textpm} 127 cm(3) for PTVFDG and 114 {\textpm} 73 cm(3) for PTVHX4. The smaller PTVHX4 overlapped on average 87 {\textpm} 16\% with PTVFDG. Prescribed dose was escalated to 87 {\textpm} 10 Gy for PTVprim, 107 {\textpm} 20 Gy for PTVFDG, and 117 {\textpm} 15 Gy for PTVHX4, with comparable doses to the relevant organs-at-risk (OAR). Treatment plans are available online (https://www.cancerdata.org/10.1016/j.radonc.2015.07.013).
CONCLUSIONS: Dose escalation based on metabolic sub-volumes, hypoxic sub-volumes and the entire tumour is feasible. Highest dose was achieved for hypoxia plans, without increasing dose to OAR. For most patients, boosting the metabolic sub-volume also resulted in boosting the hypoxic volume, although to a lower dose, but not vice versa.
}, keywords = {Aged, Aged, 80 and over, Carcinoma, Non-Small-Cell Lung, Cell Hypoxia, Female, Humans, Lung Neoplasms, Male, Middle Aged, Positron-Emission Tomography, Radiotherapy Dosage, Radiotherapy Planning, Computer-Assisted}, issn = {1879-0887}, doi = {10.1016/j.radonc.2015.07.013}, author = {Even, Aniek J G and van der Stoep, Judith and Zegers, Catharina M L and Reymen, Bart and Troost, Esther G C and Lambin, Philippe and van Elmpt, Wouter} } @article {56, title = {A Validated Prediction Model for Overall Survival From Stage III Non-Small Cell Lung Cancer: Toward Survival Prediction for Individual Patients.}, journal = {Int J Radiat Oncol Biol Phys}, volume = {92}, year = {2015}, month = {2015 Jul 15}, pages = {935-44}, abstract = {PURPOSE: Although patients with stage III non-small cell lung cancer (NSCLC) are homogeneous according to the TNM staging system, they form a heterogeneous group, which is reflected in the survival outcome. The increasing amount of information for an individual patient and the growing number of treatment options facilitate personalized treatment, but they also complicate treatment decision making. Decision support systems (DSS), which provide individualized prognostic information, can overcome this but are currently lacking. A DSS for stage III NSCLC requires the development and integration of multiple models. The current study takes the first step in this process by developing and validating a model that can provide physicians with a survival probability for an individual NSCLC patient.
METHODS AND MATERIALS: Data from 548 patients with stage III NSCLC were available to enable the development of a prediction model, using stratified Cox regression. Variables were selected by using a bootstrap procedure. Performance of the model was expressed as the c statistic, assessed internally and on 2 external data sets (n=174 and n=130).
RESULTS: The final multivariate model, stratified for treatment, consisted of age, gender, World Health Organization performance status, overall treatment time, equivalent radiation dose, number of positive lymph node stations, and gross tumor volume. The bootstrapped c statistic was 0.62. The model could identify risk groups in external data sets. Nomograms were constructed to predict an individual patient{\textquoteright}s survival probability (www.predictcancer.org). The data set can be downloaded at https://www.cancerdata.org/10.1016/j.ijrobp.2015.02.048.
CONCLUSIONS: The prediction model for overall survival of patients with stage III NSCLC highlights the importance of combining patient, clinical, and treatment variables. Nomograms were developed and validated. This tool could be used as a first building block for a decision support system.
}, keywords = {Age Factors, Aged, Analysis of Variance, Antineoplastic Combined Chemotherapy Protocols, Carboplatin, Carcinoma, Non-Small-Cell Lung, Chemoradiotherapy, Cisplatin, Deoxycytidine, Etoposide, Female, Gemcitabine, Humans, Lung Neoplasms, Male, Middle Aged, Models, Statistical, Neoplasm Staging, Nomograms, Probability, Radiotherapy Dosage, Regression Analysis, Severity of Illness Index, Sex Factors, Vinblastine, Vinorelbine}, issn = {1879-355X}, doi = {10.1016/j.ijrobp.2015.02.048}, author = {Oberije, Cary and De Ruysscher, Dirk and Houben, Ruud and van de Heuvel, Michel and Uyterlinde, Wilma and Deasy, Joseph O and Belderbos, Jose and Dingemans, Anne-Marie C and Rimner, Andreas and Din, Shaun and Lambin, Philippe} } @article {41, title = {Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach.}, journal = {Nat Commun}, volume = {5}, year = {2014}, month = {2014 Jun 03}, pages = {4006}, abstract = {Human cancers exhibit strong phenotypic differences that can be visualized noninvasively by medical imaging. Radiomics refers to the comprehensive quantification of tumour phenotypes by applying a large number of quantitative image features. Here we present a radiomic analysis of 440 features quantifying tumour image intensity, shape and texture, which are extracted from computed tomography data of 1,019 patients with lung or head-and-neck cancer. We find that a large number of radiomic features have prognostic power in independent data sets of lung and head-and-neck cancer patients, many of which were not identified as significant before. Radiogenomics analysis reveals that a prognostic radiomic signature, capturing intratumour heterogeneity, is associated with underlying gene-expression patterns. These data suggest that radiomics identifies a general prognostic phenotype existing in both lung and head-and-neck cancer. This may have a clinical impact as imaging is routinely used in clinical practice, providing an unprecedented opportunity to improve decision-support in cancer treatment at low cost.
}, keywords = {Adenocarcinoma, Carcinoma, Non-Small-Cell Lung, Carcinoma, Squamous Cell, Female, Head and Neck Neoplasms, Humans, Lung Neoplasms, Male, Multimodal Imaging, Phenotype, Positron-Emission Tomography, Prognosis, Tomography, X-Ray Computed, Tumor Burden}, issn = {2041-1723}, doi = {10.1038/ncomms5006}, author = {Aerts, Hugo J W L and Velazquez, Emmanuel Rios and Leijenaar, Ralph T H and Parmar, Chintan and Grossmann, Patrick and Carvalho, Sara and Bussink, Johan and Monshouwer, Ren{\'e} and Haibe-Kains, Benjamin and Rietveld, Derek and Hoebers, Frank and Rietbergen, Michelle M and Leemans, C Ren{\'e} and Dekker, Andre and Quackenbush, John and Gillies, Robert J and Lambin, Philippe} } @article {46, title = {Externally validated HPV-based prognostic nomogram for oropharyngeal carcinoma patients yields more accurate predictions than TNM staging.}, journal = {Radiother Oncol}, volume = {113}, year = {2014}, month = {2014 Dec}, pages = {324-30}, abstract = {PURPOSE: Due to the established role of the human papillomavirus (HPV), the optimal treatment for oropharyngeal carcinoma is currently under debate. We evaluated the most important determinants of treatment outcome to develop a multifactorial predictive model that could provide individualized predictions of treatment outcome in oropharyngeal carcinoma patients.
METHODS: We analyzed the association between clinico-pathological factors and overall and progression-free survival in 168 OPSCC patients treated with curative radiotherapy or concurrent chemo-radiation. A multivariate model was validated in an external dataset of 189 patients and compared to the TNM staging system. This nomogram will be made publicly available at www.predictcancer.org.
RESULTS: Predictors of unfavorable outcomes were negative HPV-status, moderate to severe comorbidity, T3-T4 classification, N2b-N3 stage, male gender, lower hemoglobin levels and smoking history of more than 30 pack years. Prediction of overall survival using the multi-parameter model yielded a C-index of 0.82 (95\% CI, 0.76-0.88). Validation in an independent dataset yielded a C-index of 0.73 (95\% CI, 0.66-0.79. For progression-free survival, the model{\textquoteright}s C-index was 0.80 (95\% CI, 0.76-0.88), with a validation C-index of 0.67, (95\% CI, 0.59-0.74). Stratification of model estimated probabilities showed statistically different prognosis groups in both datasets (p<0.001).
CONCLUSION: This nomogram was superior to TNM classification or HPV status alone in an independent validation dataset for prediction of overall and progression-free survival in OPSCC patients, assigning patients to distinct prognosis groups. These individualized predictions could be used to stratify patients for treatment de-escalation trials.
}, keywords = {Adult, Aged, Aged, 80 and over, Carcinoma, Squamous Cell, Chemoradiotherapy, Disease-Free Survival, Female, Head and Neck Neoplasms, Humans, Male, Middle Aged, Neoplasm Staging, Nomograms, Oropharyngeal Neoplasms, Papillomaviridae, Polymerase Chain Reaction, Predictive Value of Tests, Prognosis, Reproducibility of Results, Severity of Illness Index, Sex Factors, Squamous Cell Carcinoma of Head and Neck, Treatment Outcome}, issn = {1879-0887}, doi = {10.1016/j.radonc.2014.09.005}, author = {Rios Velazquez, Emmanuel and Hoebers, Frank and Aerts, Hugo J W L and Rietbergen, Michelle M and Brakenhoff, Ruud H and Leemans, Ren{\'e} C and Speel, Ernst-Jan and Straetmans, Jos and Kremer, Bernd and Lambin, Philippe} } @article {39, title = {International data-sharing for radiotherapy research: an open-source based infrastructure for multicentric clinical data mining.}, journal = {Radiother Oncol}, volume = {110}, year = {2014}, month = {2014 Feb}, pages = {370-374}, abstract = {Extensive, multifactorial data sharing is a crucial prerequisite for current and future (radiotherapy) research. However, the cost, time and effort to achieve this are often a roadblock. We present an open-source based data-sharing infrastructure between two radiotherapy departments, allowing seamless exchange of de-identified, automatically translated clinical and biomedical treatment data.
}, keywords = {Clinical Trials as Topic, Data Mining, Humans, Information Dissemination, Neoplasms, Radiotherapy}, issn = {1879-0887}, doi = {10.1016/j.radonc.2013.11.001}, author = {Roelofs, Erik and Dekker, Andre and Meldolesi, Elisa and van Stiphout, Ruud G P M and Valentini, Vincenzo and Lambin, Philippe} } @article {33, title = {Benefits of a clinical data warehouse with data mining tools to collect data for a radiotherapy trial}, journal = {Radiotherapy and Oncology}, volume = {108}, year = {2013}, month = {Jan-07-2013}, pages = {174 - 179}, issn = {01678140}, doi = {10.1016/j.radonc.2012.09.019}, author = {Erik Roelofs and Persoon, Lucas and Nijsten, Sebastiaan and Wiessler, Wolfgang and Andr{\'e} Dekker and P. Lambin} } @article {34, title = {Cardiac comorbidity is an independent risk factor for radiation-induced lung toxicity in lung cancer patients}, journal = {Radiotherapy and Oncology}, volume = {109}, year = {2013}, month = {Jan-10-2013}, pages = {100 - 106}, issn = {01678140}, doi = {10.1016/j.radonc.2013.08.035}, url = {http://linkinghub.elsevier.com/retrieve/pii/S0167814013004350}, author = {Nalbantov, Georgi and Kietselaer, Bas and Vandecasteele, Katrien and Cary Oberije and Maaike Berbee and Esther G C Troost and Anne-Marie C. Dingemans and Baardwijk, Angela van and Smits, Kim and Andr{\'e} Dekker and Johan Bussink and Dirk De Ruysscher and Lievens, Yolande and P. Lambin} } @article {47, title = {Prognostic value of metabolic metrics extracted from baseline positron emission tomography images in non-small cell lung cancer.}, journal = {Acta Oncol}, volume = {52}, year = {2013}, month = {2013 Oct}, pages = {1398-404}, abstract = {BACKGROUND: Maximum, mean and peak SUV of primary tumor at baseline FDG-PET scans, have often been found predictive for overall survival in non-small cell lung cancer (NSCLC) patients. In this study we further investigated the prognostic power of advanced metabolic metrics derived from intensity volume histograms (IVH) extracted from PET imaging.
METHODS: A cohort of 220 NSCLC patients (mean age, 66.6 years; 149 men, 71 women), stages I-IIIB, treated with radiotherapy with curative intent were included (NCT00522639). Each patient underwent standardized pre-treatment CT-PET imaging. Primary GTV was delineated by an experienced radiation oncologist on CT-PET images. Common PET descriptors such as maximum, mean and peak SUV, and metabolic tumor volume (MTV) were quantified. Advanced descriptors of metabolic activity were quantified by IVH. These comprised five groups of features: absolute and relative volume above relative intensity threshold (AVRI and RVRI), absolute and relative volume above absolute intensity threshold (AVAI and RVAI), and absolute intensity above relative volume threshold (AIRV). MTV was derived from the IVH curves for volumes with SUV above 2.5, 3 and 4, and of 40\% and 50\% maximum SUV. Univariable analysis using Cox Proportional Hazard Regression was performed for overall survival assessment.
RESULTS: Relative volume above higher SUV (80\%) was an independent predictor of OS (p = 0.05). None of the possible surrogates for MTV based on volumes above SUV of 3, 40\% and 50\% of maximum SUV showed significant associations with OS [p (AVAI3) = 0.10, p (AVAI4) = 0.22, p (AVRI40\%) = 0.15, p (AVRI50\%) = 0.17]. Maximum and peak SUV (r = 0.99) revealed no prognostic value for OS [p (maximum SUV) = 0.20, p (peak SUV) = 0.22].
CONCLUSIONS: New methods using more advanced imaging features extracted from PET were analyzed. Best prognostic value for OS of NSCLC patients was found for relative portions of the tumor above higher uptakes (80\% SUV).
}, keywords = {Aged, Carcinoma, Non-Small-Cell Lung, Female, Fluorodeoxyglucose F18, Humans, Lung Neoplasms, Male, Neoplasm Staging, Positron-Emission Tomography, Prognosis, Radiopharmaceuticals, Radiotherapy Planning, Computer-Assisted, Radiotherapy, Image-Guided, Tumor Burden}, issn = {1651-226X}, doi = {10.3109/0284186X.2013.812795}, author = {Carvalho, Sara and Leijenaar, Ralph T H and Velazquez, Emmanuel Rios and Oberije, Cary and Parmar, Chintan and van Elmpt, Wouter and Reymen, Bart and Troost, Esther G C and Oellers, Michel and Dekker, Andre and Gillies, Robert and Aerts, Hugo J W L and Lambin, Philippe} } @article {35, title = {The prognostic value of temporal in vitro and in vivo derived hypoxia gene-expression signatures in breast cancer}, journal = {Radiotherapy and Oncology}, volume = {102}, year = {2012}, month = {Jan-03-2012}, pages = {436 - 443}, issn = {01678140}, doi = {10.1016/j.radonc.2012.02.002}, url = {http://linkinghub.elsevier.com/retrieve/pii/S016781401200059X}, author = {Starmans, Maud H.W. and Chu, Kenneth C. and Haider, Syed and Nguyen, Francis and Seigneuric, Renaud and Magagnin, Michael G. and Koritzinsky, Marianne and Kasprzyk, Arek and Boutros, Paul C. and Wouters, Bradly G. and P. Lambin} } @article {51, title = {A semiautomatic CT-based ensemble segmentation of lung tumors: comparison with oncologists{\textquoteright} delineations and with the surgical specimen.}, journal = {Radiother Oncol}, volume = {105}, year = {2012}, month = {2012 Nov}, pages = {167-73}, abstract = {PURPOSE: To assess the clinical relevance of a semiautomatic CT-based ensemble segmentation method, by comparing it to pathology and to CT/PET manual delineations by five independent radiation oncologists in non-small cell lung cancer (NSCLC).
MATERIALS AND METHODS: For 20 NSCLC patients (stages Ib-IIIb) the primary tumor was delineated manually on CT/PET scans by five independent radiation oncologists and segmented using a CT based semi-automatic tool. Tumor volume and overlap fractions between manual and semiautomatic-segmented volumes were compared. All measurements were correlated with the maximal diameter on macroscopic examination of the surgical specimen. Imaging data are available on www.cancerdata.org.
RESULTS: High overlap fractions were observed between the semi-automatically segmented volumes and the intersection (92.5{\textpm}9.0, mean{\textpm}SD) and union (94.2{\textpm}6.8) of the manual delineations. No statistically significant differences in tumor volume were observed between the semiautomatic segmentation (71.4{\textpm}83.2 cm(3), mean{\textpm}SD) and manual delineations (81.9{\textpm}94.1 cm(3); p=0.57). The maximal tumor diameter of the semiautomatic-segmented tumor correlated strongly with the macroscopic diameter of the primary tumor (r=0.96).
CONCLUSIONS: Semiautomatic segmentation of the primary tumor on CT demonstrated high agreement with CT/PET manual delineations and strongly correlated with the macroscopic diameter considered as the "gold standard". This method may be used routinely in clinical practice and could be employed as a starting point for treatment planning, target definition in multi-center clinical trials or for high throughput data mining research. This method is particularly suitable for peripherally located tumors.
}, keywords = {Algorithms, Humans, Lung Neoplasms, Multimodal Imaging, Positron-Emission Tomography, Tomography, X-Ray Computed}, issn = {1879-0887}, doi = {10.1016/j.radonc.2012.09.023}, author = {Rios Velazquez, Emmanuel and Aerts, Hugo J W L and Gu, Yuhua and Goldgof, Dmitry B and De Ruysscher, Dirk and Dekker, Andre and Korn, Ren{\'e} and Gillies, Robert J and Lambin, Philippe} } @article {36, title = {Preclinical evaluation and validation of [18F]HX4, a promising hypoxia marker for PET imaging}, journal = {Proceedings of the National Academy of Sciences}, volume = {108}, year = {2011}, month = {Jun-08-2013}, pages = {14620 - 14625}, issn = {0027-8424}, doi = {10.1073/pnas.1102526108}, url = {http://www.pnas.org/cgi/doi/10.1073/pnas.1102526108}, author = {L. J. Dubois and Lieuwes, N. G. and Marco H. M. Janssen and Peeters, W. J. M. and Windhorst, A. D. and Walsh, J. C. and H. C. Kolb and M. C. Oellers and Johan Bussink and van Dongen, G. A. M. S. and van der Kogel, A. and P. Lambin} } @article {37, title = {PET imaging of hypoxia using [18F]HX4: a phase I trial}, journal = {European Journal of Nuclear Medicine and Molecular Imaging}, volume = {37}, year = {2010}, month = {Jan-08-2010}, pages = {1663 - 1668}, abstract = {\
Download the images using\ these instructions\ and this DOI :\ 10.1007/s00259-010-1437-x
\
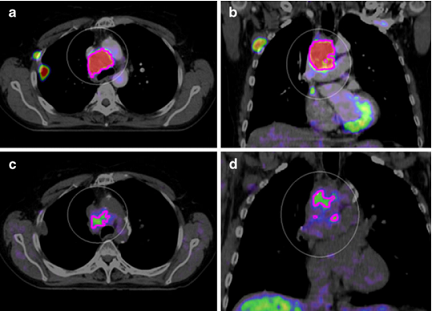
Background and purpose
Non-invasive PET imaging of tumour hypoxia could help in the selection of those patients who could benefit from chemotherapy or radiation with specific antihypoxic treatments such as bioreductive drugs or hypoxic radiosensitizers. In this phase I trial, we aimed to determine the toxicity of [18F]HX4, a member of the 2-nitroimidazole family, at different dose levels. The secondary aim was to analyse image quality related to the HX4 dose and the timing of imaging.
Methods
Patients with a histologically proven solid cancer without curative treatment options were eligible for this study. A study design with two dose steps was used in which a single dose of a maximum of 222MBq (step 1) or 444 MBq (step 2) [18F]HX4 was injected. Toxicity was scored on day 0 and on days 3 and 7 after injection, according to the CTCAE 3.0 scoring system. PET/CT images of the largest tumour site were acquired 30, 60 and 120 min after injection.
Results
Six patients with stage IV carcinoma were included, four with non-small-cell lung carcinoma, one with thymus carcinoma, and one with colon carcinoma. No toxicity was observed in any of the patients at either dose level. The median tumour to muscle ratio 120 min after injection was 1.40 (range 0.63{\textendash}1.98).
Conclusion\
The findings of this study showed that [18F] HX4 PET imaging for the detection of hypoxia is not associated with any toxicity. Imaging was successful; however, future trials are needed to determine the optimal image parameters.